Microscale Transport, Interfaces, and Biology
Our lab investigates transport phenomena at the micro- and nanoscale, where fluid mechanics, interfacial physics, and confinement govern the behavior of multiphase systems, particles, and living cells. We study droplets, emulsions, nanoparticles, and active matter to uncover how flow unsteadiness, surface forces, and material properties control deformation, mixing, clustering, and transport. By integrating microfabrication, advanced imaging, theoretical analysis, and high-fidelity simulations, we build mechanistic frameworks that connect fundamental physics to predictive design. This foundation enables the development of microfluidic platforms for molecular diagnostics, particle characterization, and controlled biological assays. Our work extends to single-cell electroporation and mechanobiology, probing how electrical and mechanical cues regulate membrane dynamics, gene delivery, and cellular responses. We also explore microbial and amoeboid behavior in complex mechanical environments to inform disease mechanisms and therapeutic strategies. Below listed are summaries of our varied research areas bridging fundamental microscale transport with impactful technologies in diagnostics, biopharmaceutical analysis, and biomedical engineering.
Mechanics and Control of Bacterial Attachment, Colonization, and Biofilms
Our research seeks to understand how mechanical forces, fluid flow, and electric fields collectively regulate bacterial behavior, surface colonization, and community formation, with a particular focus on Pseudomonas aeruginosa. This organism exhibits a rapid transition from planktonic motility to surface attachment through mechanosensitive pathways that integrate hydrodynamic cues with intracellular signaling. By recreating pulsatile flow environments characteristic of physiological systems, we investigate how time-varying shear, confinement, and nutrient transport influence early colonization dynamics, population-level feedback, and the stability of initial attachment under realistic flow conditions.
Building on this foundation, we examine how physical fields can be leveraged both to interrogate and to control bacterial–surface interactions. Using AC electrokinetic forcing, we explore how externally applied fields reorganize bacterial populations and disrupt the progression toward biofilm formation on abiotic surfaces. In parallel, we probe the very early-time mechanics of bacterial adhesion, resolving the force-mediated interactions that precede irreversible attachment and trigger adaptive genetic and phenotypic responses. Together, this work advances a unified mechanobiological framework for understanding and regulating bacterial colonization at the intersection of fluid mechanics, electromechanics, and infection-relevant environments.
Relevant Projects:-
Hydrodynamic mediation of bacterial adhesion kinetics by pulsatile flow profiles.
Electrohydrodynamic control of bacterial surface adhesion using applied electric fields and electro-osmotic slip.
POC: Shubhonil Sarkar
Single-Cell Electromechanics in Eukaryotic Systems
Through a collaboration with HP Labs, we aim to refine gene delivery by tailoring electrical pulses to individual cells. With access to a state-of-the-art single-cell electroporation device, we can investigate how electrical and mechanical stresses influence membrane permeabilization, repair kinetics, and transfection efficiency at the single-cell level. This careful method could improve cell survival, reduce damage from electroporation, and enhance the effectiveness of gene editing. In the long term, the project may contribute to personalized cancer therapies by enabling safer and more effective engineering of T-cells for immunotherapy. Currently, we are conducting experiments with CHO-K1 cells, with a plan to include Jurkat cells and HEK293T cells in the near future. We aim to gain a mechanistic understanding of pore formation at a single-cell level, leading to applications towards rare-cell therapy.
A collaboration between our lab and the RICE Research Group at Purdue University investigates how Acanthamoeba, a free-living amoeba responsible for the severe corneal infection Acanthamoeba keratitis, responds to mechanical cues in its environment. By culturing Acanthamoeba on substrates with varying stiffness and using high-resolution 3D imaging tools, we examine how substrate stiffness, surface topology, and solid–air interfaces influence encystment, trophozoite motility, and surface interactions. Understanding these mechanobiological factors may yield new insights into how the amoeba invades and degrades corneal tissue, ultimately informing better strategies to prevent or treat this vision-threatening disease.
Relevant Projects:-
Understanding the biophysical mechanisms of electroporation and optimizing transfection efficiency.
A Mechanobiological Study on Acanthamoeba Keratitis.
POC: Sazid Noor Rabi

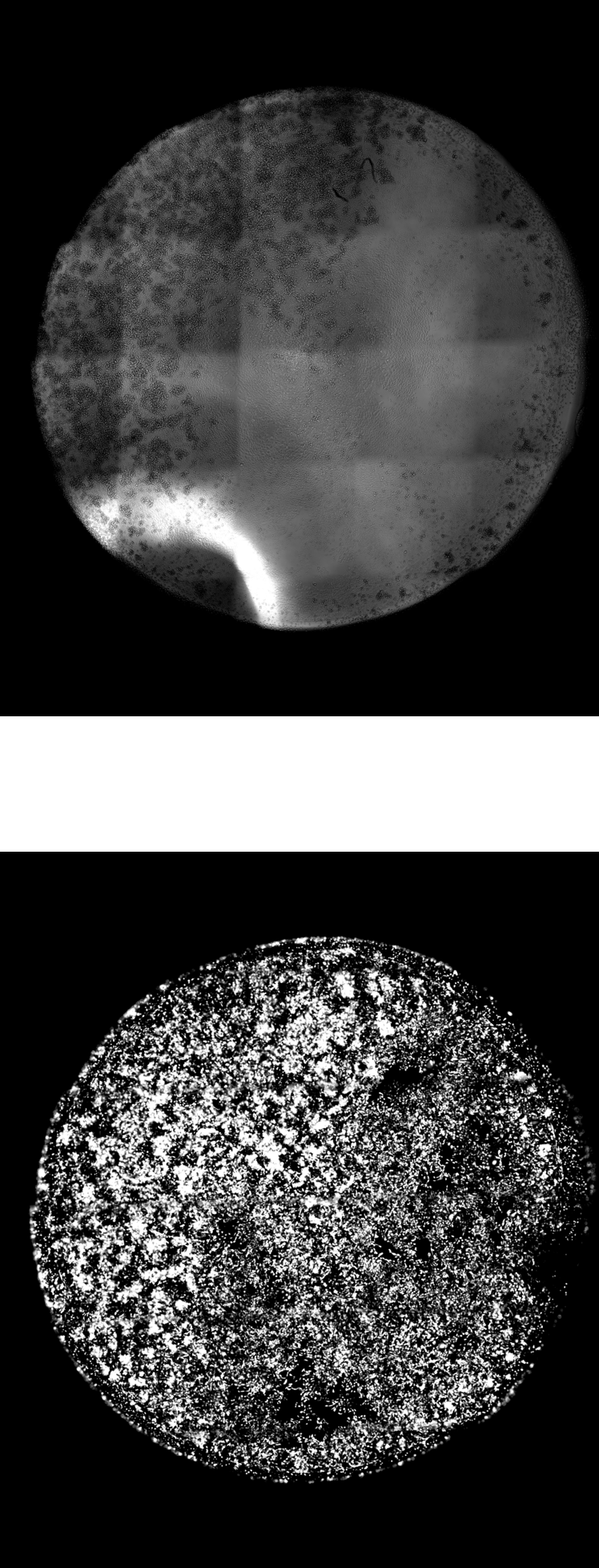

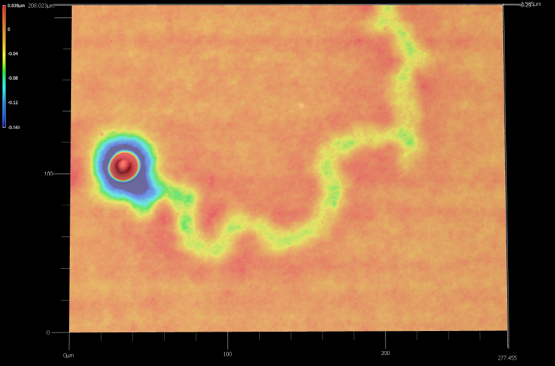
Characterization of Subvisible Particles in Therapeutic Formulations

Relevant Projects:-
Automated subvisible particle counting, characterization, and ingredient-level identification in viscous biologics using 3D profilometry and correlative hyperspectral imaging.
Contactless, label-free real-time chemical characterization using Isotachophoresis integrated with Raman spectroscopy.
POC: Luren Wang
One of our thrust areas is characterization of subvisible particles under different fluid environments: how subvisible particles form, deform, and interact within the tightly confined and highly viscous environments characteristic of modern pharmaceutical formulations. In these systems, drugs share different compositions, viscosities, interfacial behavior, etc. Our work involves developing platforms that preserve native microstructure while enabling accurate counting and ingredient-level chemical identification. To discern subvisible particles, without using labeled or destructive preparation steps, remains a challenge. The research aims to construct a trustworthy representation of particulate behavior in complex biological formulations and to provide a bridge between formulation conditions and aggregate formation pathways.
This foundation in particle imaging supports our broader goal of advancing analytical tools for biopharmaceutical development. As biology becomes more concentrated and more complex, analytical workflows must transition from single-channel, low-throughput instruments to platforms that are robust, automated, and compatible with small sample volumes. Ultimately, the aim is to transform how the community detects, interprets, and manages subvisible particles in therapeutics —linking fundamental understanding of particulate formation with technologies that improve drug robustness, manufacturability, and safety.
Theoretical Active Matter Dynamics in Unsteady Environments
We investigate the fundamental question of how motile bacteria and other microswimmers are transported and concentrated in oscillatory flows. Oscillatory flows are ubiquitous across natural, biological, and engineered systems at a wide range of scales, ranging from cardiovascular and tidal environments to pipelines, medical devices, and filtration networks. Yet the transport behavior of active particles in oscillatory flows remains poorly understood. Motivated by this gap, our work combines theoretical analysis with high-fidelity Langevin dynamics simulations to determine how flow unsteadiness, shear, and cell motility interact to shape migration patterns and spatial redistributions of active particles in oscillatory environments. By identifying the physical mechanisms that give rise to preferential clustering and depletion, we aim to establish a mechanistic framework for predicting transport in active suspensions subjected to unsteady forcing. The work has broad real-world relevance, informing the design of microfluidic platforms for diagnostics and particle manipulation, improving transport predictions in medical and environmental flows, and guiding strategies to mitigate biofilm formation in industrial and biomedical systems.
Relevant Projects:-
Transport and preferential concentration of microswimmers in oscillatory flows.
POC: Raghav Ram